Research Blog

When you’re caring for someone with Prader-Willi syndrome (PWS), you become an expert at navigating complex medical and behavioral needs. But even with the best care, emergencies can still happen. A new study, funded by FPWR and led by Dr. James Luccarelli, examines why individuals with PWS visit...
Source: FPWR Blog
|
Published: May 5 2026 - 06:30 AM

We are pleased to announce the recipients of FPWR's first round of grants in 2026, totaling more than $1 million in funding to support innovative research and bold initiatives in Prader-Willi syndrome (PWS) and Schaaf-Yang syndrome (SYS).
Source: FPWR Blog
|
Published: May 5 2026 - 06:15 AM
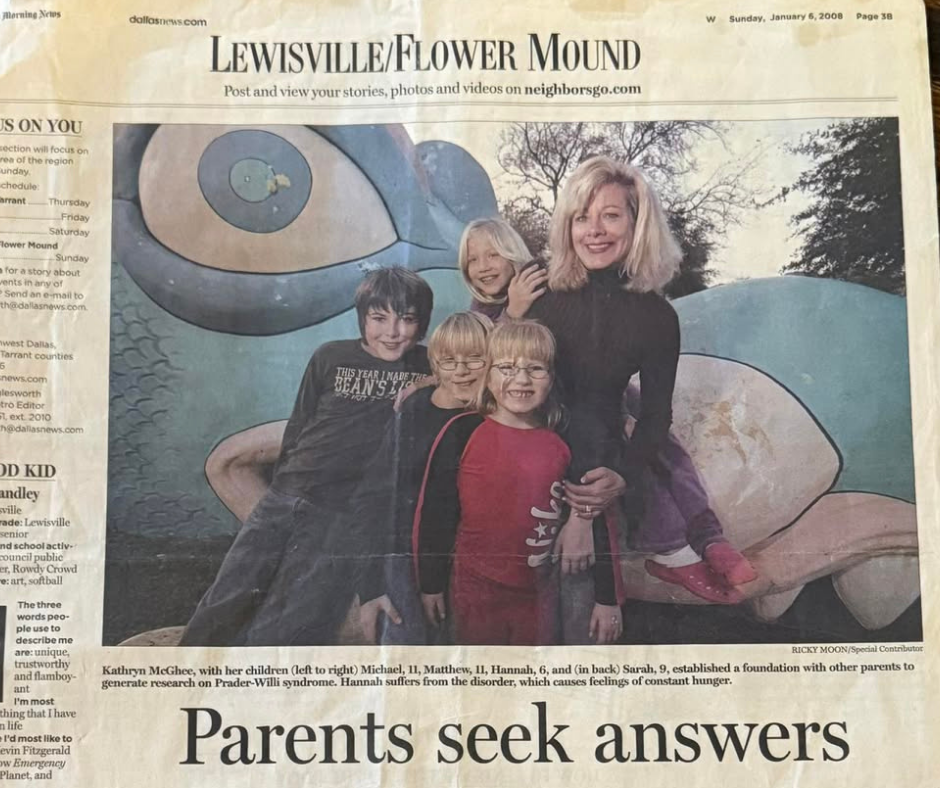
In January 2008, The Dallas Morning News published an article titled “Parents Seek Answers.” At the center of the story was Kathryn McGhee, a mother navigating the daily realities of Prader-Willi syndrome while also helping shape a growing research movement that would become the Foundation for...
Source: FPWR Blog
|
Published: May 5 2026 - 05:45 AM

FPWR maintains the latest clinical trial information so that you can stay abreast of trial opportunities. Some trials require in-person visits, while others can be completed remotely. Some trials are testing new drugs, while others are intervention (non-drug) or natural history studies. Details of...
Source: FPWR Blog
|
Published: May 5 2026 - 12:30 AM

Families raising a child with Prader-Willi syndrome know how difficult temper outbursts can be. In the study “The Characteristics of Temper Outbursts in Prader-Willi Syndrome,” Drs. Rice, Einfeld, and Woodcock, experts in the field of PWS, took a closer look at these behaviors to better understand...
Source: FPWR Blog
|
Published: April 4 2026 - 05:45 AM










