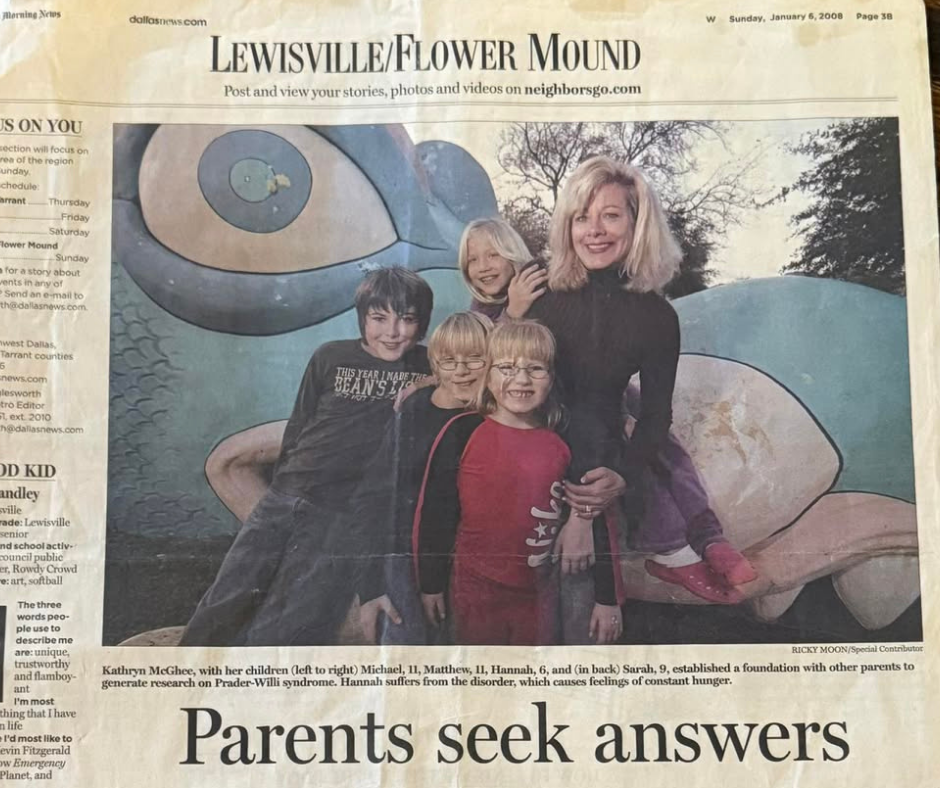This article includes personal experiences shared by Lisa Matesevac and clinical guidance from Dr. Harold van Bosse, MD
Scoliosis is a common orthopedic complication in Prader-Willi syndrome (PWS), affecting the majority of individuals at some point during infancy, childhood, or adolescence. Scoliosis in PWS is not the same as the more familiar forms of scoliosis. Dif...
When you’re caring for someone with Prader-Willi syndrome (PWS), you become an expert at navigating complex medical and behavioral needs. But even with the best care, emergencies can still happen. A new study, funded by FPWR and led by Dr. James Lucc...
Topics: Clinical Issues, Research
A new phase 2 study is evaluating CSTI-500, an investigational medication being developed to address some of the most challenging symptoms of PWS, including hyperphagia and behavioral dysregulation. Researchers will study the safety, tolerability, an...
We are pleased to announce the recipients of FPWR's first round of grants in 2026, totaling more than $1 million in funding to support innovative research and bold initiatives in Prader-Willi syndrome (PWS) and Schaaf-Yang syndrome (SYS).
Topics: Research, Schaaf-Yang Syndrome
Aardvark Therapeutics has announced that the U.S. Food and Drug Administration (FDA) has placed a full clinical hold on its investigational new drug application (IND) for ARD-101 related to the Company’s previously announced voluntary pause.
When people think about Prader-Willi syndrome (PWS), conversations often focus on the challenges. But just as important—and too often overlooked—are the strengths and positive attributes that define individuals with PWS.
An expert review, written by Drs. Miller, Shoemaker, Salehi, and Corletto, offers practical clinical guidance for the use of diazoxide choline extended-release (DCCR/VYKAT™ XR), the first FDA-approved treatment for hyperphagia in Prader-Willi syndrom...
In January 2008, The Dallas Morning News published an article titled “Parents Seek Answers.” At the center of the story was Kathryn McGhee, a mother navigating the daily realities of Prader-Willi syndrome while also helping shape a growing research m...
Topics: Stories of Hope, Research, Parents, hopefull