Prader-Willi syndrome (PWS) is a complex disorder, and, to date, supportive care and growth hormone therapy are the only treatments available. The rarity of PWS, the fragmentation of experts and stakeholders involved at each stage of therapeutic development, and limited funding add layers of complexity in the development of safe and efficacious PWS therapies.
Prader-Willi syndrome (PWS) is a complex disorder, and, to date, supportive care and growth hormone therapy are the only treatments available. The rarity of PWS, the fragmentation of experts and stakeholders involved at each stage of therapeutic development, and limited funding add layers of complexity in the development of safe and efficacious PWS therapies.
Therapeutic development is a risky process that often requires more than 10 years and billions of dollars to complete. Unfortunately, 90 percent of drugs fail due to a lack of effect in patients.
The research plan for PWS needs to be comprehensive and proactive and include the right programs and tools to overcome these challenges and fill existing gaps. FPWR’s grant program and other research initiatives developed over the last decade have greatly advanced our basic understanding of both the genetic and molecular mechanisms of PWS, opening avenues for therapeutic development.
FPWR’s 5-year research plan for PWS comes at a crucial moment to accelerate the development of therapies by acting at each stage of the therapeutic development pathway.
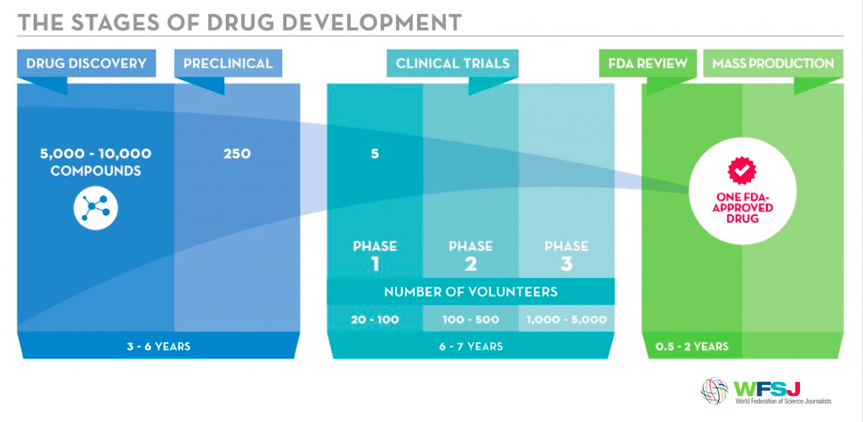
The Three Main Stages of Drug Development
The therapeutic development process must proceed through three main stages to produce a drug that is safe and efficacious and that has fulfilled all regulatory requirements. FPWR has developed research programs and tools to “de-risk” drug development along each stage of the development pathway. (The graphic below is courtesy the World Federation of Science Journalists.)
The first stage of drug development involves drug discovery: identifying a target for which a drug can be developed. Target identification relies on identifying the biological mechanism(s) that is not working correctly in PWS.
FPWR has developed three programs/tools to address critical gaps at the drug discovery stage:
- Through the Critical Knowledge Program, FPWR will identify new therapeutic targets by understanding how the loss of the PWS genes and the changes in the brains of those with PWS lead to the characteristics of PWS.
- The PWS Cellular Network aims at developing PWS cell models and assays that are critical for screening drugs in a high-throughput fashion to identify potential compounds for drug development.
- The Global PWS Registry, designed to help us better understand the full spectrum of PWS characteristics, also guides the identification of targets that are relevant in human trials.
Preclinical Research
Once a target has been identified and a drug has been selected, it needs to be tested in an animal model that recapitulates the mechanisms the drug is intended to repair. This stage, called the preclinical stage, is required before a drug candidate can be tested in humans.
FPWR has developed two programs to address the preclinical challenges:
- The Preclinical Animal Network Program aims to increase the stringency of drug selection at the preclinical stage by validating preclinical mouse models and developing a drug screening platform.
- The New Animal Models Program aims at developing novel PWS animal models to expand the range of current studies, most of which have been conducted in mice. The development of larger-animal models, such as in pigs, which have a physiology closer to that of humans, could bring additional information or potentially be better predictors of responses in humans than rodent models. In each case, it will be necessary to choose the best model for a specific application.
Clinical Trials
Once a drug candidate has been proven to be safe and efficacious in PWS animal models, it can be tested in clinical trials in which the drug is evaluated in a subset of patients within a regulated and controlled environment. This stage typically consists of three phases (I, II, and III) to test the safety and efficacy of a drug candidate. If successful, the regulatory agencies provide a market authorization for the sponsor organization to market the drug.
Clinical trials for PWS face several challenges, including a limited number of available patients, the lack of objective measures to assess drug efficacy, the lack of knowledge on the natural progression of the disease, and the unknown levels of risk patients and caregivers are willing to take for the benefit of a novel therapeutic. Having identified these challenges and the resources necessary to overcome them, FPWR is actively taking steps to address these challenges and facilitate the completion of clinical trials.
The Global PWS Registry allows rapid identification of patients who meet the eligibility requirements of a given trial and are willing to participate. The PWS Clinical Trial Consortium addresses clinical trial challenges, which include identifying better outcome measures, understanding the natural progression of the disease, and incorporating the patient and caregiver's voices in the benefit/risk assessment of therapeutic options through a collaborative and international network of stakeholders from industry, academia, and patient organizations. In addition, the PWS Biobank is intended to support the development of biological markers so we may have more objective tools to measure drug efficacy.
FPWR's Role in Facilitating Therapeutic Development
Drug development requires the input of multiple stakeholders and experts—patients, researchers, industry, and government—and FPWR is fostering and guiding collaborations between the multiple stakeholders and experts at each stage of the drug development pathway.
Through our research plan, FPWR is de-risking drug development by developing critical programs that no other stakeholders are willing to fund or develop while positioning our syndrome to be a more attractive disease target for industry (who have the means to take a drug all the way through clinical trials) and increasing the likelihood of developing a successful PWS therapies.